Digital Lab Solutions AI Product Menu
PathAI is a leading IMS provider with deep expertise in algorithm development and with real-world experience from implementing AI in its own routine pathology laboratory. Thus, we offer a curated, multi-partner digital pathology ecosystem that gives labs seamless access to world-class algorithms that are fully integrated into AISight’s single viewer and workflow functionalities.
Workflow Optimization
ArtifactDetect
Automated detection of artifacts using action-oriented classes to support quality monitoring.
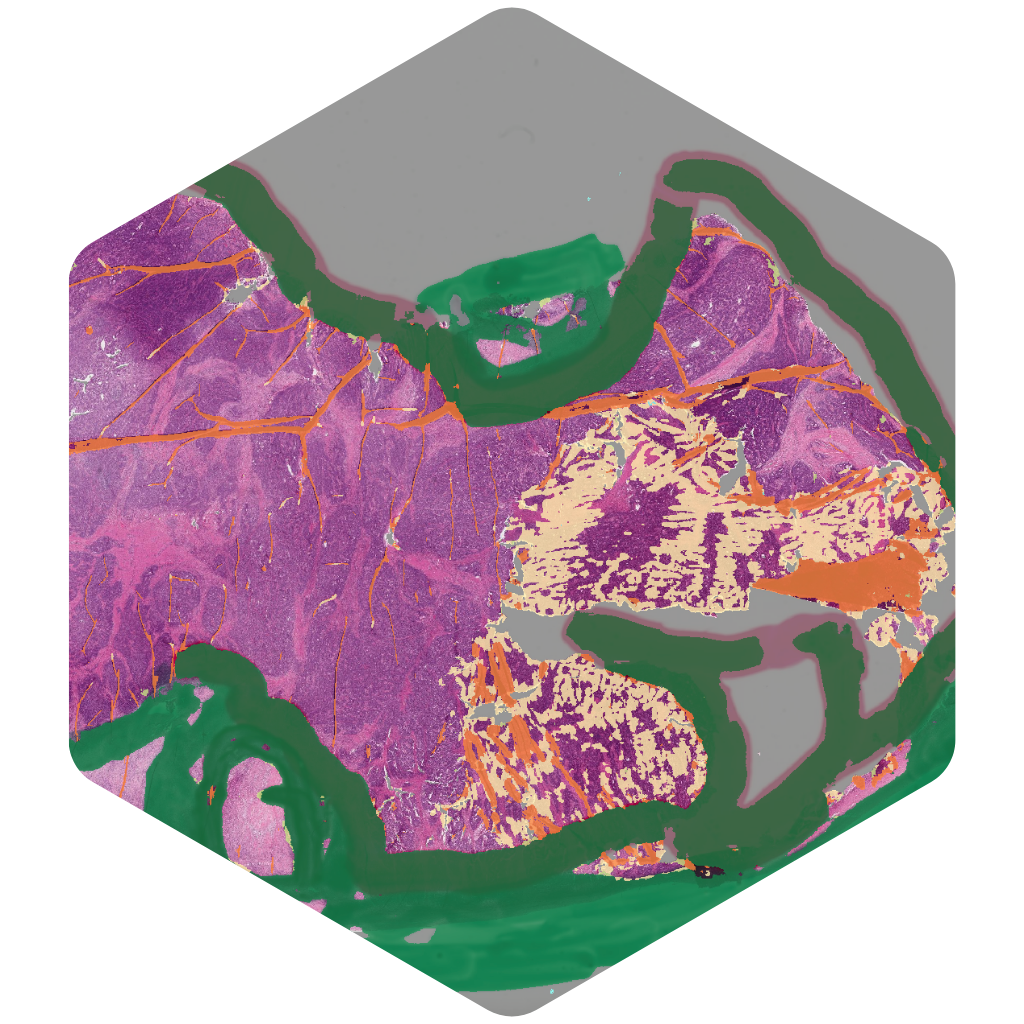
TumorDetect
Automated detection of tumor across all common carcinomas for case prioritization and case triaging.
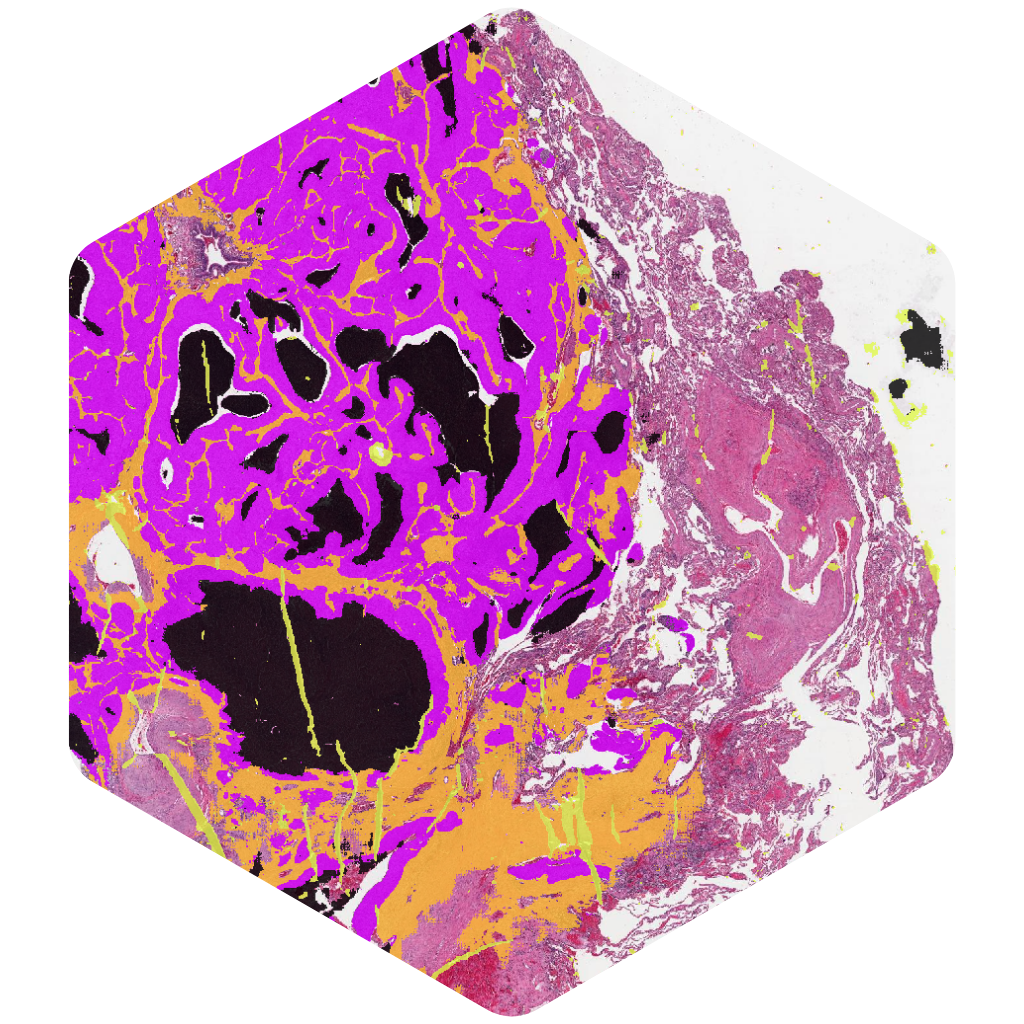
AIM-TumorCellularity
Automate tumor-cellularity assessment for molecular testing workflows.
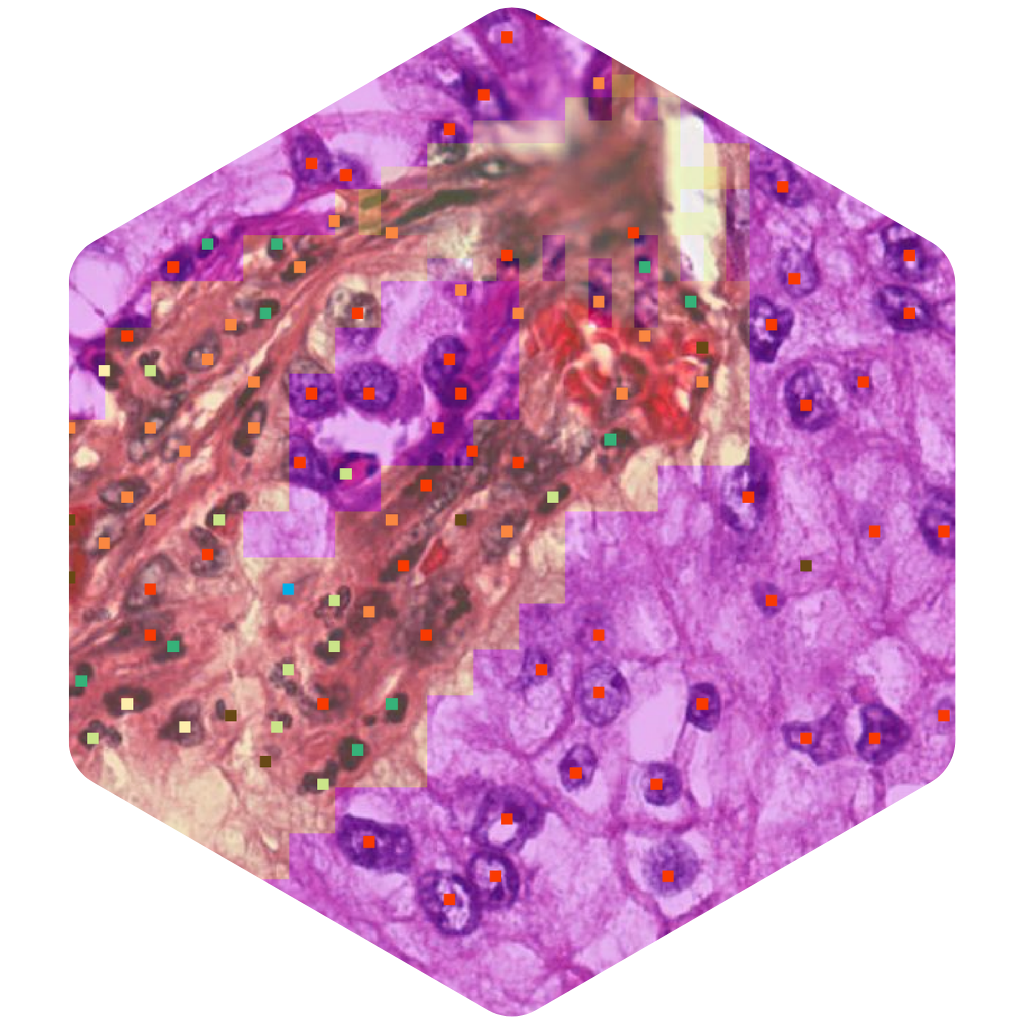
Biomarker Quantification
AIM-PD-L1
Detect and quantify PD-L1+ tumor and immune cells. Available for biopharma and lab research use.

AIM-IHC Breast Panel
AI-Based Measurement Panel of Key Breast Cancer Biomarkers — HER2, ER, PR, and Ki-67.
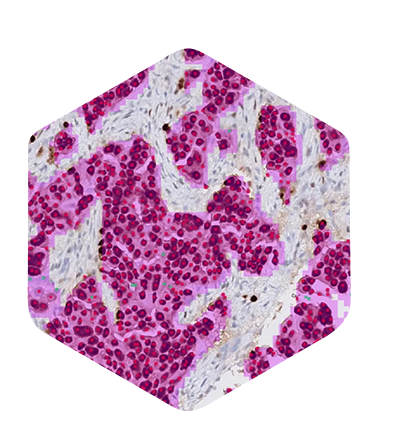
AIM-MASH+
Automated and reproducible, AI-powered ordinal and continuous MASH CRN score component grades and fibrosis stage.
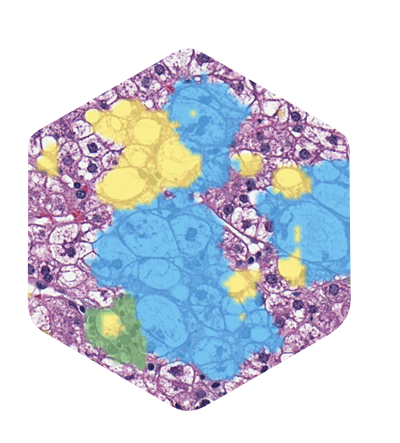
PathAssist
PathAssist Derm
AI-assisted histopathology tool for specimen orientation, prioritization, and measurement.
PathAI AI Partners
Deep Bio
DeepDx Prostate, is a CE-IVD-marked diagnostic aid designed to enhance the precision of prostate cancer assessment in digital pathology.
DoMore Diagnostics
Histotype Px® Colorectal is an outcome prediction marker for stage II and III colorectal adenocarcinoma that informs the decision of whether to provide adjuvant chemotherapy following surgical resection. It is CE-IVD-marked in EU.
MindPeak
Mindpeak delivers CE-IVD-marked AI solutions for breast cancer biomarker analysis (HER2, ER/PR, Ki-67) and lung PD-L1, complemented by RUO assays for prostate, gastric, esophageal, urothelial
cancers, and onychomycosis in dermatopathology.
Paige
Paige’s AI applications alleviate time and resource pressures for pathologists. Paige’s AI suite includes solutions for Prostate, Breast, Colon, and Pan-Cancer.
Primaa
Primaa offers AI-powered diagnostic tools such as Cleo Breast (CE-IVDR) and Cleo Skin (RUO) designed to detect and accurately quantify cancer biomarkers, lesion types, dimensions, margins, mitoses, and perineural invasion.
Stratipath
Stratipath provides the first CE-IVD marked AI-based solution for prognostic risk profiling of breast cancer using routine histopathology slides.
Visiopharm
Visiopharm offers a full breast panel of APPs (ER, PR, Ki-67, Metastasis Detection, and HER2) as well as an APP for non-small cell lung cancer (PD-L1). These APPs are cleared under IVDR in EU and UK and are RUO in the rest of world.

ArtifactDetect is a workflow tool available on AISight® and AISight® Dx. AISight is for Research Use Only. Not for use in diagnostic procedures. AISight Dx is FDA-cleared for primary diagnosis in the US and is CE‑IVD–marked for primary diagnosis in the EEA, UK, and Switzerland.
TumorDetect, AIM-TumorCellularity, AIM-PD-L1, AIM-IHC Breast Panel, AIM-MASH and PathAssist Derm are for Research Use Only. Not for use in diagnostic procedures.
