AI-based MASH Scoring
PathAI has developed AI-based measurement (AIM) tools that provide Clinical Research Network (CRN) MASLD Activity Score (MAS) component grades and fibrosis stages.

AIM-MASH AI Assist1 delivers accurate and reproducible histologic assessment with pathologist review
AIM-MASH AI Assist aids pathologists in MASH CRN scoring to reduce the impact of reader variability and capture drug effect in clinical trials. This tool is currently under review by the FDA and qualified by the EMA to “aid a single central pathologist for enrollment/inclusion of patients into clinical phase 2 and phase 3 trials in MASH” and for the “evaluation of study outcomes (primary or secondary) in case this is intended to be based on histology evaluation.2
.webp?width=100&height=100&name=Check-List-13%20(1).webp)
Improve Scoring Confidence
Reduce the impact of scoring variability in MASH clinical trials and research.

Streamlined Workflows
Streamline trial workflows and conserve valuable resources by accelerating time-to-result with an AI-assisted single reader.
Robust Development
Trained on a vast and diverse dataset of >5,900 biopsies from across six different clinical trials.
AIM-MASH AI Assist Specifications
- Intended Use: Research Use Only
- Indications: MASH
- Scanners: Leica Aperio® AT2
- Inputs: 1 H&E and 1 Masson’s Trichrome image
- Outputs: Pathologist AI-assisted ordinal MASH CRN scores; AI-alone ordinal MASH CRN scores
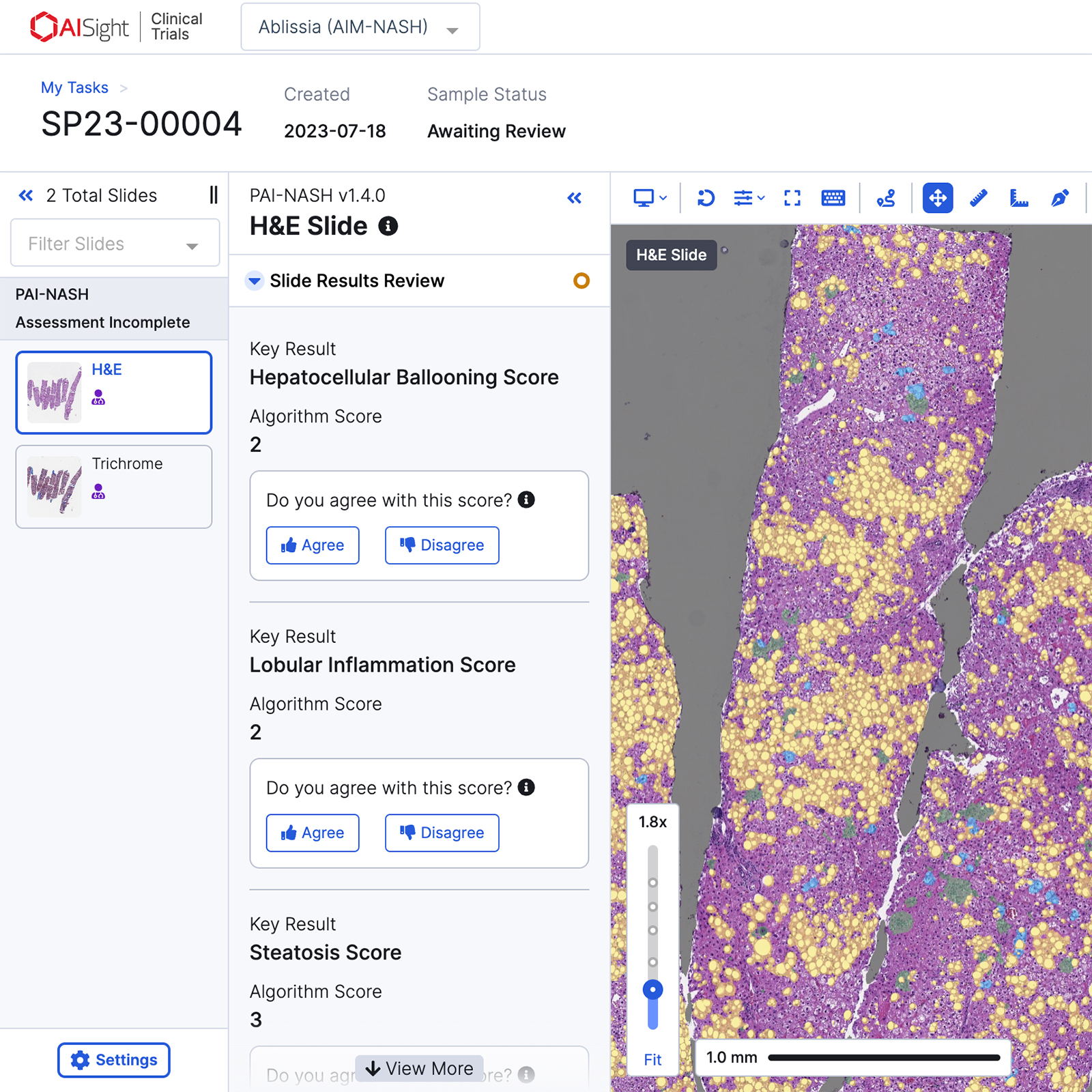

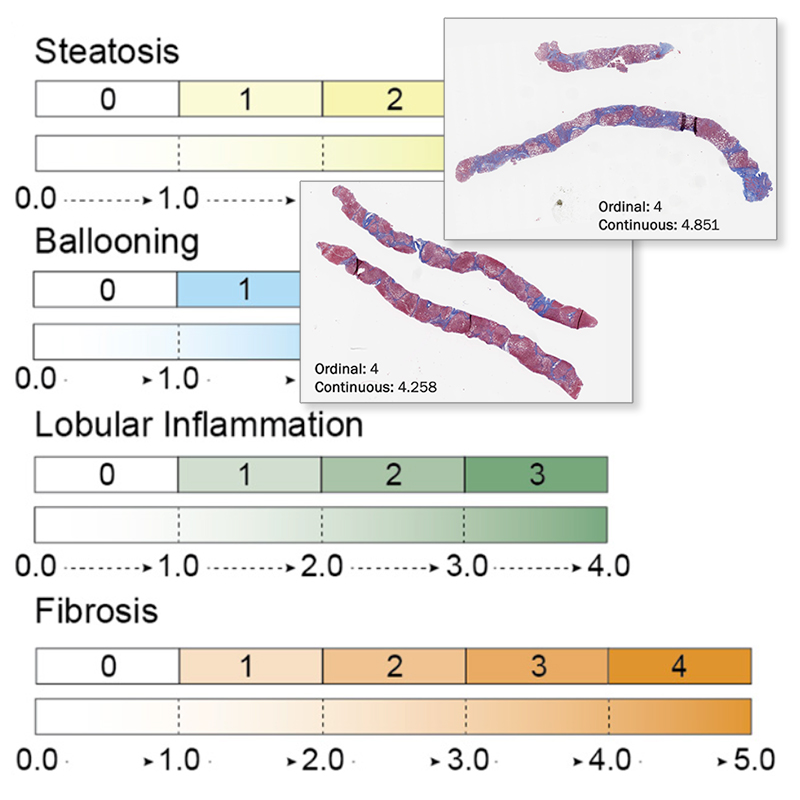
AIM-MASH+2 provides a nuanced and deeper understanding of histological changes within CRN grades/stages
AIM-MASH+ outputs AI-alone ordinal and continuous MASH CRN scores to drive more nuanced and deeper understanding of changes within CRN grades/stages.

Robust, High-Resolution Read Outs
AI-based continuous scoring of MASH histologic features can reveal smaller degrees of change which can lead to improved precision medicine strategies.

Directly Comparable
Direct association between ordinal and continuous CRN score measures enables granularity without compromising.
.webp?width=100&height=100&name=Check-List-13%20(1).webp)
Added Assurance
AI-only ordinal scores provide objective, independent and reproducible output to test or confirm findings from pathologist read.
AIM-MASH+ Specifications
- Intended Use: Research Use Only
- Indications: MASH
- Scanners: Leica Aperio® AT2
- Inputs: 1 H&E and 1 Masson’s Trichrome image
- Outputs: AI-alone ordinal MASH CRN scores; AI-derived continuous MASH CRN scores; Artifact model QC features
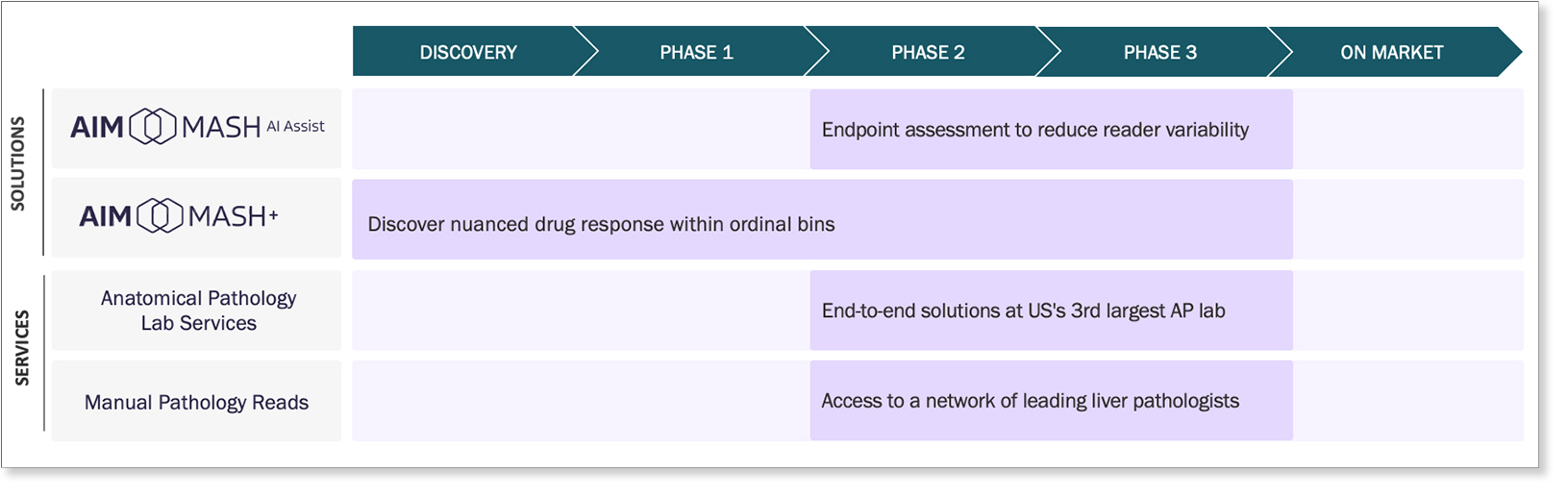
PathAI support throughout various stages of the MASH therapeutic development pipeline
Resources
Connect With Our Team
Contact us to explore opportunities to accelerate your MASH Clinical Program.
1 AIM-MASH AI Assist is qualified as a tool in the EU and as a DDT in the US for use in MASH clinical trials. AIM-MASH AI Assist is not for use in diagnostic procedures.
2 AIM-MASH+ is for Research Use Only and is not for use in diagnostic procedures.
