
AISight Consults in AISight® Dx IMS
One platform for end-to-end consult management.
PathAI’s AISight® Dx IMS platform supports consult workflow capabilities through the AISight Consults workflow, enabling institutions to send, receive, review, and report on consult cases within a unified IMS experience, without adding a separate consult tool. Designed to help move beyond fragmented consult processes, AISight Consults can help reduce reliance on glass slide shipment and manual coordination while supporting faster turnaround time and real-time collaboration between referring and consulting pathologists.
Less shipping. Less manual coordination. More connected consult workflows.
Digital consults built into the IMS workflow
Send, receive, review, and report on consult cases within the existing AISight Dx IMS workflow, without adding a separate consult tool.
Less reliance on glass shipment
Digital case submission, slide viewing, reporting, and communication help reduce the burden of packaging, shipping, tracking and coordinating glass slide consults.
Scalable consult intake and review
Organized worklists, pathologist assignment rules, and structured reporting help consulting institutions manage consult volume more efficiently.
Faster access to subspecialty expertise
Referring institutions can submit cases digitally, receive updates, access reports, and collaborate through annotations and real-time co-viewing.
A practical entry point into digital pathology
For labs not yet fully digitized, AISight Dx supports consult workflows through manual or bulk upload, without requiring full LIS integration from day one.

Unlike standalone consult tools, the AISight Dx supports consultation workflows within the platform your lab already uses, helping reduce operational complexity while supporting implementation, onboarding, and ongoing optimization.
Two audiences. One integrated workflow.
AISight Dx supports both sides of the consult relationship, helping consulting institutions manage inbound volume and referring institutions reduce physical glass transit within a single, connected platform.
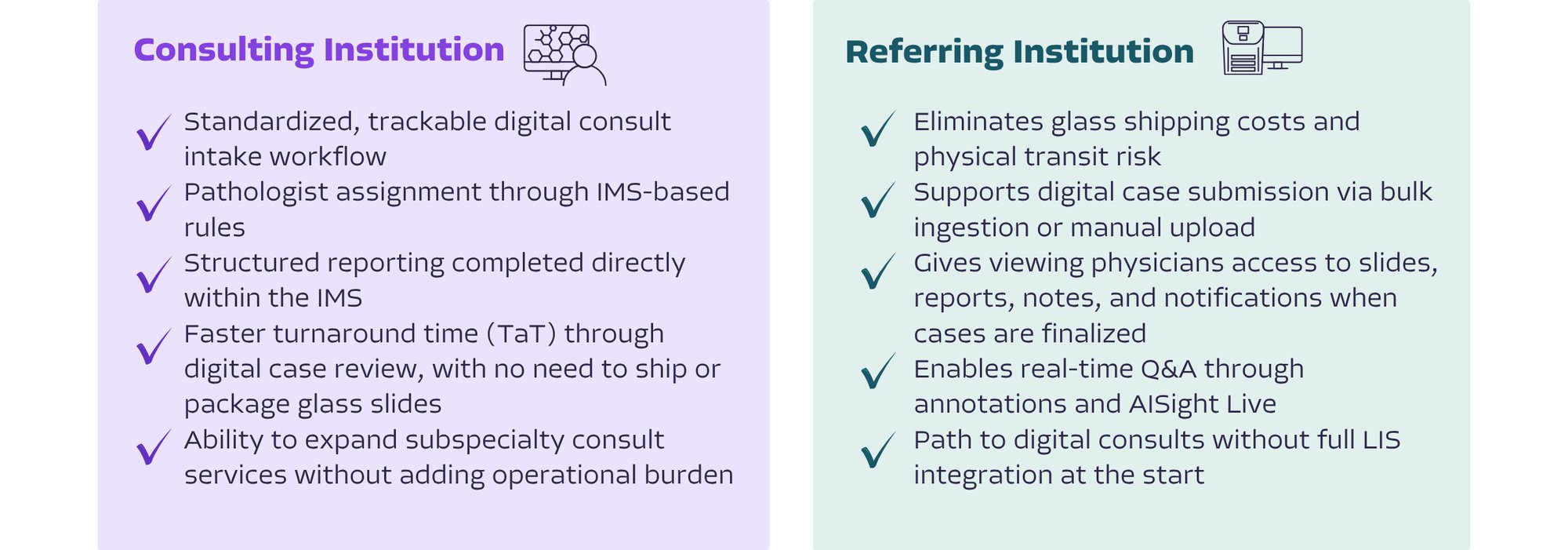
See how AISight Dx supports digital consult workflows.
CASE STUDY
AI-Assisted Titer Selection in Early Assay Development
- PathAI deployed IHC Explore on prostate cancer specimens stained with a novel, in-development assay
- IHC Explore quantifies staining intensity at single-cell resolution, enabling rapid assay characterization and titer optimization
- Continuous staining intensity measurement provides added value for next-generation biomarkers and precision medicine strategies
Schedule a free demo today
fuel your pathology workflows & research with AISight Dx
*AISight® Dx is FDA-cleared for primary diagnosis in the US and CE-IVD-marked for primary diagnosis in the EEA, UK, and Switzerland.
